Do surface coatings influence cell migration?
Experiments show that surface coatings play an important role in cell movement
When performing cell migration experiments, a perennial question is: what surface coatings should be used to culture a particular cell type? Scientists working in Cancer Research, Wound Healing, or Drug Discovery utilize cell cultures to make important experiments and advance our understanding of biological mechanisms. In particular, assays for cell migration enable characterization of conditions and substances that influence movement of cells. For example, scientists using the OrisTM Cell Migration Assays successfully identified proteins, mRNA and antioxidants that inhibit migration of tumor cells.
Each cell type requires specific conditions for optimal growth, so it is important to choose the right surface to culture each cell type. For example, fibroblasts produce and inhabit the connective tissue of the body; thus we may hypothesize that surfaces and scaffolds that are rich in collagen and other fibers (components of connective tissue) are better suited for culturing fibroblasts. This means that collagen-coated surfaces are a good choice when culturing fibroblasts for cell migration assays. But does the choice of surface coating influence cell migration?
In new experiments, scientists at Platypus Technologies demonstrate that the choice of surface coating for cell culture may influence the results for cell migration experiments.
Oris™ Cell Migration Assays use a 96-well plate with “stopper” barriers that create a central cell-free Detection Zone for cell migration experiments. Removing the stoppers allows the cells to migrate into the Detection Zone at the center of each well:

In this experiment, the scientists used a cell line called HT1080, which are fibroblasts isolated from a malignant human tumor. The cells were incubated on the OrisTM Cell Migration Assays that had wells coated with five different bioactive surfaces: Tissue Culture, Collagen I, Fibronectin, Poly-L-Lysine, and Basement Membrane Extract (BME). Following cell attachment, the OrisTM stoppers were removed to permit cell migration into the detection zone. The cells were then imaged following 24 hours of incubation without the OrisTM stoppers. Representative images from this experiment are shown below:
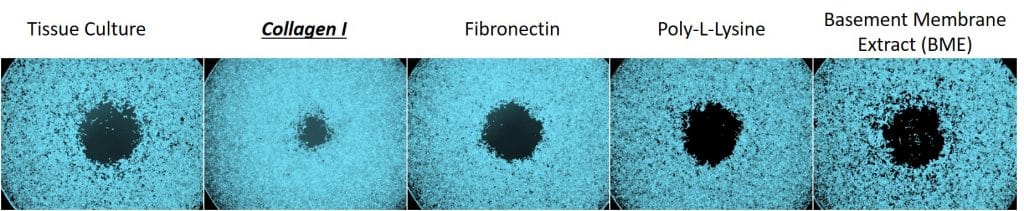
Through visual inspection of these images, we observe that the cells cultured on Collagen I migrate deeper into the detection zone, compared to cells cultured on other bioactive surfaces. There is no noticeable difference in the migration of the cells cultured on tissue culture, fibronectin, poly-l-lysine or basement membrane extract.
Twelve (12) different wells from each plate were imaged prior and after cell migration, and the area of the cell-free zone was measured using ImageJ. The difference in the area of the cell-free zone pre- and post- migration was used to calculate the percentage area closure of the assay. These measurements are presented in the graph below, which show unequivocally that HT1080 cells exhibit higher migration rates in Collagen I than in any other surface coating.
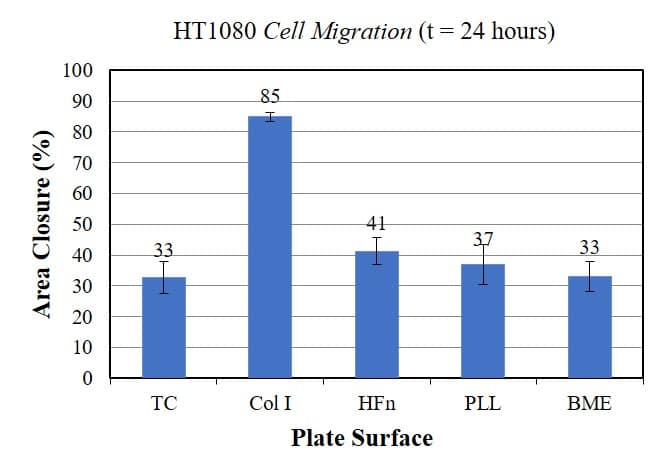
In conclusion, the choice of surface coating has a strong effect on the cell migration of HT1080 cells. Cells cultured in collagen I exhibit the fastest rate of cell migration, compared to cells cultured in other surfaces. These results demonstrate that the choice of surface coatings plays an important role on the measured cell migration.
Learn more about Oris Cell Migration Assays.
Additional References:
Stochastic and Heterogeneous Cancer Cell Migration: Experiment and Theory