Cell Invasion Assay (3D)
Oris™ : A SIMPLE PLATFORM FOR PUBLICATION-READY DATA
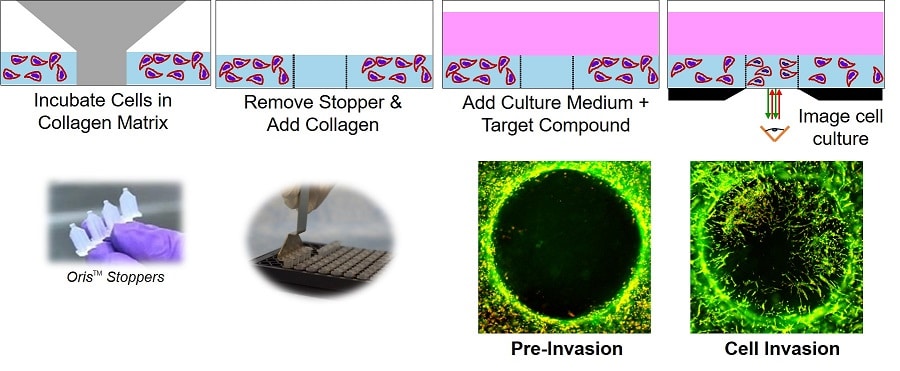
In the Oris™ 3D Cell Invasion Assay, stoppers are used to create a central collagen-containing Detection Zone for cell invasion experiments.
Cells are suspended in a thick layer of collagen surrounding a circular collagen Detection Zone. After incubation, invading cells move into the detection zone, where they can be unambiguously quantified using a microscope or a plate-reader.
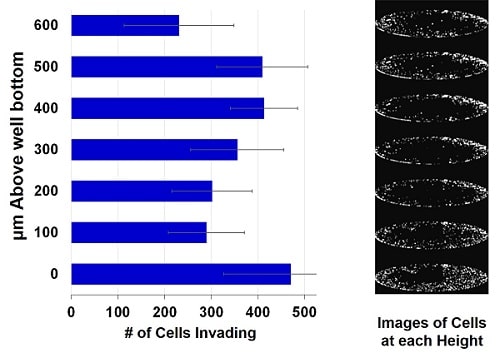
Above: Image analysis of the cells embedded in collagen confirm 3D distribution of cells.
Features & Benefits
- Monitor cell invasion entirely in 3D
- Oris™ platform enables you to distinguish different cell behavior in 2D cell migration and 3D cell invasion assay
- Physiologically Relevant: No artificial membranes or inserts required
- Versatile: Supports real-time monitoring or end-point analysis
- Easy Analysis: Invasion can be quantified using image analysis or plate readers; no elaborate cell tracking software needed
Applications
Cell invasion assays are important tools for cancer research. Scientists studying cancer perform cell invasion assays to measure the cell movement through an extracellular matrix. By varying the conditions of the cell culture (e.g. adding new drugs), scientists can identify conditions that prevent or accelerate cell invasion. Through these studies, new treatments for cancer can be identified for potential therapeutic use.
Literature
Check out our poster at the ASCB Conference HERE.
Find Protocol HERE.
Examples of Studies that used Oris™ 3D Invasion Assay:
- D. Zhuo et al. Inhibitor compounds for endothelial-to-mesenchymal transition. J. of Cancer 2024, LINK
- X. Zhang et al. “Actin cytoskeleton reorganization of hepatic stellate” Cell Adh Migr, 2019, LINK.
- L. Bertier et al. “Downregulation of invadopodium formation” Biomed. Pharm. 2018, LINK
- P. Masuzzo et al. “Analysis of high-throughput single-cell migration data” Sci. Reps. 2017, LINK
Ordering Information
| SKU | Product Description | |
|---|---|---|
| EIA1 | Oris™ 3D Invasion Assay, 1-pack | BUYNOW |
| EIA3 | Oris™ 3D Invasion Assay, 3-pack | BUY NOW |
| CI48 | Oris™ Collagen I (Rat tail) 5 mg/mL | BUY NOW |
Oris™ is a trademark of Platypus Technologies, LLC. Oris™ technology is protected by US patents 7,842,499, 8,268,614 and 8,512,974 and pending applications.